Image: https://www.chemistryworld.com/opinion/what-is-an-atom/3007562.article
Atomic theory is the theory that everything is made of atoms (which are made of smaller particles, neutrons, protons, and electrons) that are slightly different between elements in regards to the number of each subatomic particle they contain.
For a quick timeline of atomic theory and the model of the atom click here
For a quick timeline of atomic theory and the model of the atom click here
Dalton’s Theory:
Proposed the concept of the mole and recognized that compounds form from the union of the atoms of different elements. He also assigned the first atomic weights of elements, giving a relative atomic weight of one to hydrogen
Proposed the concept of the mole and recognized that compounds form from the union of the atoms of different elements. He also assigned the first atomic weights of elements, giving a relative atomic weight of one to hydrogen
Dalton's Model of the Atom
Image: https://www.tes.com/lessons/m4AVPHjEGTesdA/atomic-models-through-time
Image: https://www.tes.com/lessons/m4AVPHjEGTesdA/atomic-models-through-time
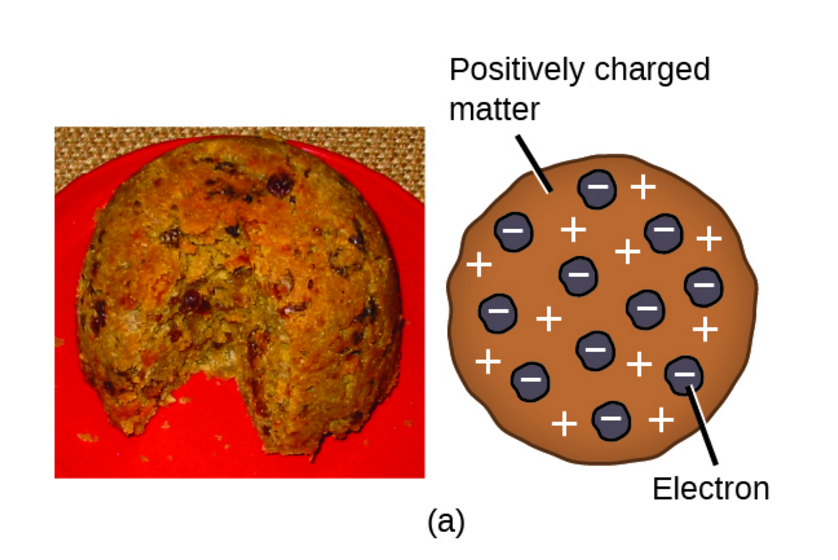
Plum Pudding Model:
J.J. Thomson identified electrons in his cathode ray tube experiment. This atomic model suggested that atoms consist of a positive mass (the pudding) with the smaller, negative electrons (raisins in the pudding) stuck throughout the positive mass.
J.J. Thomson identified electrons in his cathode ray tube experiment. This atomic model suggested that atoms consist of a positive mass (the pudding) with the smaller, negative electrons (raisins in the pudding) stuck throughout the positive mass.
The plum pudding model of the atom (right) and the plum pudding dessert (left)
Image: https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/history-of-atomic-structure/a/discovery-of-the-electron-and-nucleus
Image: https://www.khanacademy.org/science/chemistry/electronic-structure-of-atoms/history-of-atomic-structure/a/discovery-of-the-electron-and-nucleus
Ernst Rutherford: Proposed the nuclear atom after discovering protons during his gold-foil experiment. He also proposed the existence of neutrons.
Bohr’s Model
Niels Bohr proposed that electrons orbit around the atom’s nucleus in “electron shells” With a certain amount of electrons in each layer of the atom's shell.
Werner Heisenberg proposed the principle of indeterminancy—you cannot know both the position and velocity of a particle (like an electron) at the same time.
Niels Bohr proposed that electrons orbit around the atom’s nucleus in “electron shells” With a certain amount of electrons in each layer of the atom's shell.
Werner Heisenberg proposed the principle of indeterminancy—you cannot know both the position and velocity of a particle (like an electron) at the same time.
Bohr's model of the atom
Image: (left) https://www.timetoast.com/timelines/quimica-su-inicio-hasta-el-dia-de-hoy (right) https://scienceconceptions.wikispaces.com/Orbiting+Electrons
Image: (left) https://www.timetoast.com/timelines/quimica-su-inicio-hasta-el-dia-de-hoy (right) https://scienceconceptions.wikispaces.com/Orbiting+Electrons
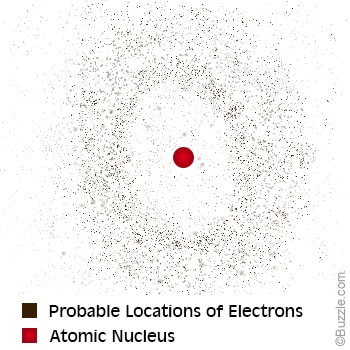
Electron Cloud
Electrons move randomly around the atom’s nucleus.
Erwin Schrodinger proposed that electrons move around protons as continuous clouds instead of in orbits and “introduced wave mechanics as a mathematical model of the atom (Buescher, Lee).”
Electrons move randomly around the atom’s nucleus.
Erwin Schrodinger proposed that electrons move around protons as continuous clouds instead of in orbits and “introduced wave mechanics as a mathematical model of the atom (Buescher, Lee).”
Electron cloud model
Image: https://sciencestruck.com/electron-cloud-model
Image: https://sciencestruck.com/electron-cloud-model
Buescher, Lee. Atomic Structure Timeline. Atomic Timeline, 2017. http://atomictimeline.net/